|
|
The carbon dioxide (C02) is a colorless, odourless , dense and little reactive gas, with spicy flavour, corrosive in the humidity presence. It forms part of the troposphere (layer of the atmosphere next to the Earth ), currently in a ratio of 350 pfm (parts for million). Its cycle in the nature is tied with the one of the oxygen.
The carbon dioxide, or carbonic anhydride is a chemical composition consisting by two atoms of oxygen and a carbon atom. The chemical representation is CO2. The carbon dioxide was discovered by the Scot Joseph Black in 1754. It is the main made up of the expelled volcanic gases during the eruption.
Without this gas the Earth would be an ice block. On the other hand, a CO2 excess hinders the exit of heat of the atmosphere, provoking a planet heating called greenhouse effect.
Also known as carbonic anhydride, the carbon dioxide is the result of the combination of two chemical elements: the carbon (C) and the oxygen (O2), and its chemical representation is CO2, what means that its molecule is composed of a carbon atom for two of oxygen. It is good for remembering that the O2 is a molecule that contains two atoms of oxygen (O) ( whose atomic number is 8 → 8 atoms and 8 protons). When it appears in molecular form, or O2, the oxygen is presented in gaseous form and constitutes 20% of the terrestrial atmosphere. He is one of the elements most important of organic chemistry, participating in excellent way in the energy cycle of being living creatures, and being essential in the cellular breath of aerobics organisms, amongst which the mammals (where the human beings are enclosed) and the plants.
EXPIRATION OF THE CARBON DIOXIDE GAS
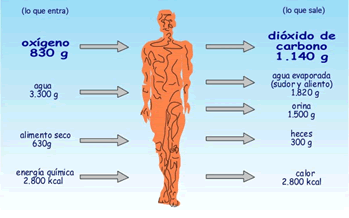
In each day, each human being inspires 830g of O2 and exhales 1140 g of CO2. That is: in the inspiration the animal absorbs oxygen and, in the expiration, it expels carbon dioxide.
 |
O que entra -
O que sai
www.urbanext.uiuc.edu/gpe_sp/images_rev/air.gif |
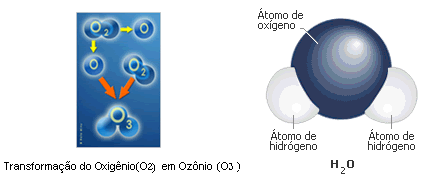
There exists, also, a molecular form consisting by three atoms of oxygen, O3, that is called ozone, whose bigger presence is finding in the stratosphere, where there are agglomerations of them , forming the ozonosphere - in conditions of quantitative normality, the ozone layer protects the Earth of the ultraviolet radiation incidence coming from the Sun. On the other hand, its reduction is lethal to the being living creatures of the terrestrial surface. There is one third molecular formula, with only one atom of oxygen, called free or phenolic oxygen (O). This last one can result of an electric load received by the O2 molecule (with all its electron layers complete), which is so divided in two molecules of free oxygen, each one with its last electron layer incomplete, therefore needing to join with another one proportionally incomplete with its same substantial characteristics or of diverse substance, to complete itself. Free oxygen is called so because it did not suffered yet any union with another atom. It is very rare to meet free oxygen in the atmosphere, because each one of these atoms is always binding to others of same or other characteristics, sharing, thus, with the other substance, pairs of electrons, to become steady and to fulfill its functions in the nature. Through “chemical reactions”, natural or induced, it is that the molecules of diverse substances are formed, where everyone bind the last layers of other free atom , in the same condition with oppositive electronic charge, sharing, both, thus, pairs of electrons, changedding themselves. As in the example below, where the arrow means “is changedded in” :
O2 + electric load → O+O , that, in turn, can form two ozone molecules if they find two molecules of the O2. If the meeting will be only with a molecule of O2 , Ozone is completed with only one atom of the O , and the other, continuing free and unstable, looks for immediately to league to another substance, equally free and unstable, as Hydrogen, H (in this case it is necessary two free Hydrogen atoms for that free Oxygen) which goes to result in a very , very know composed - the water,also named monoxide of hydrogen or hydrogen oxide, whose molecular formula is H2O {meant: a water molecule is composed for two Hydrogen atoms (H) and one atom of Oxygen (O)}. It is noticed that atoms of free Hydrogen and free Oxygen are rare in the atmosphere, therefore in the nature there will be always having chemical reactions, for a reason of a natural electric force, leaguing other free atoms or unstable substances to it.
|
( Oxygen Atom)
|
|
Convertion of Oxygen (O2) into Ozone (O3) ( Hydrogen Atom) ( Hydrogen Atom) |
Note 1 : The Hydrogen element , in pure state, with only one atom, is called Helium, very rare in our atmosphere and very frequent substance in the solar atmosphere, from there its name. In the surface of our planet, it is a fossil gas founded in natural reserves in determined wells of extration of petroliferous reserves, extremely led and volatile . It was very used in fuels, especially of the airships and, today, it is usded more for attainment of an inert atmosphere , in liquid state. Of future, one expects sufficiently to use it in the cooling of atomic reactors. It is not healthful for the inhalation by human being. When the atom of Helium loses an electron, becoming exempts, it is leagued immediately to another one in the same situation - in the case of proper Hydrogen, it forms the H2 molecule.
 |
Note 2 : determined substances transform into others through linkings. The linkings can be made of three forms:
a) the metallic ( among metals) : in a determined metal alloy, it has trend, in certain circumstances, of electron release of finish layers of each atom, making a chain of cations, dived in an electron net - applied an electric field on them, an electric chain is created;
b) ionic: in the case of metal with not metals and metals with hydrogen; the atoms of these elements change electrons of the last layer (eletrosphere) between themselves, being the receiver with more “n” electrons than the number of its protons and the giver with less “n” electrons of what its protons , always one gaining a unit electronic for that the other loses, calling itself, the first one, anion, and the other, cation. Being the loss and the profit equivalents, it has positive and negative equivalence of forces - forming a very strong electric attraction. (Electronic Formula of Lewis);
c) for sharing (covaliant) : polar, simple or dative. In the case of the CO2, it has court appointed covalency. In this case, it exists an electron in excess in the last layer of a free atom (anion) and in scarcity in another one (cation); then, the atom with more electrons than protons (electronegative), remains in the same place, creating a polarity in its respective last layer (eletrosphere), to where goes the other element, with less electrons than protons (eletropositive) - the attraction take place in relation to the other atom, passing extreme electrons to be integrant part of both the last layers (eletrospheres) of both atoms, filling thenselves mutually with their correct numbers, saying that they had been saturated. But if there is lack or surplus of electrons even though this sharing in the last layer of the eletrosphere of one or all those atoms, that operation can be repeated, binding them innumerable free atoms or innumerable other substances, until they are completely saturated (or better, till they complete their last electron layers, equaling the total number of protons and electrons of each atom), forming true chains. When it has a mutual electron donation between two atoms, has court appointed linking covaliant as in the CO2. See more in: http://luizclaudionovaes.sites.uol.com.br/ligaquim.htm
In the case of the CO2, the direction of donation is from the more eletronegative atom, the Oxygen, that has six electrons in the last layer, to the less eletronegative, the Carbon, that possess 4 electrons in its one. In the contrary to the rule, therefore - but the result, in objective character of the reaction, do not diverge of what occurs ordinary , in contrary direction. In accordance with the sharing, all are with 8 electrons in each last layer, each one supplying to the last layer of the other, 2 electrons of its last layer: none of them loses, all earn and they saturate mutually - each atom of Oxygen gains 2 electrons and the Carbon atom gains 4 electrons, 2 of each Oxygen. All are, therefore dependent between thenselves, and therefore, they continue joined ones .
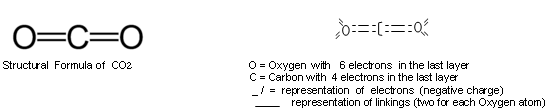 |
The CO2 is, therefore, a steady substance, product of reaction in different processes, such as, the combustion of the coal and the hydro-carbons, the fermentation of the liquids, and the breath of the human beings and the animals. In the terrestrial atmosphere, where also it is found , the normal one is that it is in weak concentration. This gas is assimilated by the plants, that for their turn operate a chemical reaction in their inside, during which occurs the steady of this stable molecule, with release of oxygen (O2) and the retention of carbon. The gaseous CO2 has a slightly irritating, colorless odor and is more weighed than the air. Its gaseous state change, when in a very cold temperature, the 78,5 ºC negative, forming the called “carbonic snow” (in this state it is very unstable to be easily able to be isolated). And, at most cold , still, it pass to the solid state, and is called “dry ice”.
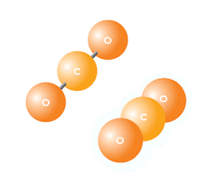 |
Representation of CO2 Molecule, where C means Carbon and ,O, Oxygen
It is composed by two Oxygen insaturated atoms and one Carbon insaturated atom
See more : www.carbonforce.net/public/index.php?option=c..
|
The CO2 is used in drinks, to give bubbling to them. And , during fires , in extinguishers, to isolate the oxygen of the fuel .
THE CARBON DIOXIDE CYCLE ON EARTH’S SURFACE
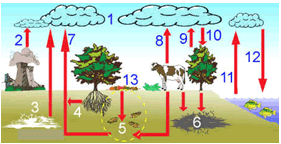 |
| 1 - Carbon in the atmosphere
2-Factories/powersthermal
3- Located deposist
4-Breath of the roots
5-Decomposition
6-Deposit of fóssiles fuels (coal, oil, natural gas…)
7-Emission of the ground and breath of the organisms
8-Breath of terrestrial animals (enclosed human beings)
9 - Breath of plants
10 - Assimilation by plants
11 - Breath of the seaweed and aquatic animals
12 - Photosyntheses of seaweed 13 - Remaining portion .
www.natureduca.com/.../cienc_ciclo_carbono .jpg
|
The oxygen breathed for the aerobic organisms, set free for the plants in the photosynthesis process, participates in the conversion of nutrients in intracellular energy.
The reduction of the oxygen level provokes the hipoxemia and the total lack causes anoxia, being able to provoke the death of the organism.
The hemoglobin is the pigment that gives the color to red globules (eritrocits) and has the vital function to distribute the oxygen for the organism.
See more :
http://pt.wikipedia.org/wiki/Oxig%C3%A9nio
http://luizclaudionovaes.sites.uol.com.br/ligaquim.htm
www.projeto-biologico.arizona.edu/.../09t2.gif
www.urbanext.uiuc.edu/gpe_sp/images_rev/air.gif
www.cricyt.edu.ar/.../images/dioxcarb.jpg
http://pt.wikipedia.org/wiki/Di%C3%B3xido_de_carbono
http://educar.sc.usp.br/licenciatura/2003/ee/Efeito_Estufa.html
http://noticias.pluc.com.br/showarticle.php?articleID=992
http://www.feiradeciencias.com.br/sala02/02_100.asp
www.juntadeandalucia.es/.../ver/09/fotosin.gif
www.somosamigosdelatierra.org/00_imagenes/inv...
www.corpohumano.hpg.ig.com.br/.../i_pulmones.jpg
|